 |
| Petroleum |
Petroleum has been known throughout historical time even before the modern industrialization. It was used mainly in battlefield especially in mortar and other fire weapon of the defensive warfare. However, it is also used in coating walls and boat hulls. Not to mentioned, the Native Americans used petroleum in the making of medicine (Seneca oil and Genesee oil) and paints. In 1815, several streets in Prague were lighted with petroleum lamps.
 |
| E . L . Drake |
The modern petroleum industry actually began in 1859, when the American oil pioneer E . L . Drake drilled a producing well on Oil Creek in Pennsylvania at place that later became Titusville. At that time, with the petroleum industry, they produced kerosene and these kerosene are used in lamps which soon replaced the whale oil lamps and candles in general use.
About Petroleum
Petroleum is a naturally occurring, yellow-to-black liquid found in geological formations beneath the Earth's surface, which is commonly refined into various types of fuels. Petroleum is formed when large quantities of dead organism, usually zooplankton and algae, are buried underneath sedimentary rock and subjected to intense pressure. Forming fossil fuels.
| Natural petroleum spring in Kornia, Slovakia |
ety has its unique mix of molecules which determines its physical and chemical properties.
In the strictest sense, petroleum includes only crude oil. However, in common usage it includes all liquid, gaseous and solid hydrocarbons. Under different temperature and pressure conditions, they appear in different form and has different usages.
| Most of the world oil are non-conventional |
| Element | Percent range |
|---|---|
| Carbon | 83 to 85% |
| Hydrogen | 10 to 14% |
| Nitrogen | 0.1 to 2% |
| Oxygen | 0.05 to 1.5% |
| Sulfur | 0.05 to 6.0% |
| Metals | < 0.1% |
Composition of Hydrocarbons in Petrol
| Hydrocarbon | Average | Range |
|---|---|---|
| Alkanes (paraffins) | 30% | 15 to 60% |
| Naphthenes | 49% | 30 to 60% |
| Aromatics | 15% | 3 to 30% |
| Asphaltics | 6% | remainder |
Petroleum is recovered mostly through oil drilling. This comes after the studies of structural geology, sedimentary basin analysis, reservoir characterization. It is then refined and separated, most easily by distillation into a large number of consumer products, from gasoline and kerosene to asphalt and chemical reagents used to make plastics and pharmaceuticals.
Fuels (Petrol & Diesel)
The most common distillation fractions of petroleum are fuels.
Fuels are used in vehicles and transportation system which has became our daily necessity.
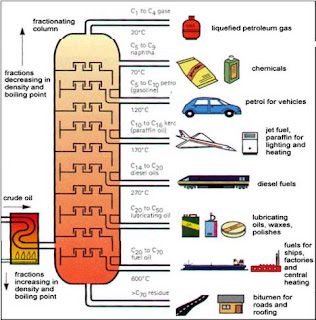 |
| Distillation fraction |
Industrial power
Oil is the major source of industrial power. While diesel and gasoline are commonly used in gas turbines to produce electricity.
Heating and lighting
Heavier oils are used in central heating plants for shops, offices as well as homes.
Heavier oils are used in central heating plants for shops, offices as well as homes.
The lighter grades of oil such as Kerosene are still used for domestic purposes.
Lubricants
All types of lubricants and greases are produced from petroleum. These lubricants are very useful for machines especially for the machines that are used in transportation and industries.
Petro-chemical industry
Both petro-chemical and chemical industries use petroleum by-products as raw material to produce chemical fertiliser, synthetic fiber, synthetic rubber, nylon, plastics, pesticides and insecticides, perfumes, dyes, paints, carbon black and sulphur, etc.
Environmental effects
As petroleum is a naturally occuring substance, its presence in the environment need not be the result of human causes such as accidents and routine activies (seismic exploration, drilling, extraction, refining and combustion).
Ocean acidification
Ocean acidification is the increase in acidity of the Earth's oceans caused by the uptake of carbon dioxide from the atmosphere. This increase in acidity inhibits life such as scallops.
Global warming
 |
| Petroleum drilling site |
Combustion of petroleum releases carbon dioxide, a greenhouse gas. Along with the burning of coal, petroleum combustion is the largest contributor to the increase in atmospheric carbon dioxide. This has cause global warming and raise in temperature on the Earth leading to melting of icebergs in North and South pole, which is extremely dangerous as it leads to rise in sea water level.
Extraction
Oil extraction is simply the removal of oil from the reservoir (oil pool). This may sounds harmless but it has causes the Earth to be damaged especially geologically.
Oil spills
 |
| Kelp after an oil spill |
Crude oil and refined fuel spills from tanker ship accidents have dramatically damaged natural ecosystems in Alaksa, the Gulf of Mexico, the Galapagos Islands, France and many other places. Oil spills at sea are generally much more damaging than those on land, since they can spread for hundreds of nautical miles in a thin oil slick which can cover beaches with a thin coating of oil. This can kill organisms such as sea birds, mammals, shellfish and other organisms it coats.
Before we end this topic here,
one advice to all the people out there.
 |
| A poster used to encourage carpooling during World War II |













